With the development of urbanization and industrialization, the problem of water pollution has attracted more and more attentions. Drinking water is normally transported to terminal consumption through pipe networks after being treated by waterworks. Despite various indicators meet the drinking water standards, the treated water may generate disinfection byproducts, soluble heavy metals, and pathogens after transporting in the pipe networks, water towers and tanks. Among these problems, the concentration of heavy metals in the outlet drinking water may exceed the limited values due to variations of physiochemical properties of water in pipes. This phenomenon would give a potential risk to residents. For example, lead ions released from lead pipes, tin coatings, brass materials, and PVC pipes can damage the nervous system, reproductive system and kidneys. Because of its high specific surface area and rich varied surface chemistry, activated carbon (AC) is an ideal material for removing multiple contaminants. Meanwhile, there are some limitations in the application of AC: (ⅰ) low efficiency and capacity of heavy metal removal; (ⅱ) the secondary contamination caused by biofilm formed on the surface of AC. The nanoscale zero-valent iron is a promising environmental functional material and is able to remove various kinds of contaminations. However, the application of nanoscale zero-valent iron is limited by its easily aggregation and surface passivation.
Therefore, the Group of Clean Energy Technology and Carbon Material (team of Pro.Wang Yin) in Institute of Urban Environment, Chinese Academy of Sciences firstly synthesized an activated carbon supported nanoscale zero-valent iron (NZVI/AC) composite at ultralow iron content but with function of safely and high-efficiently converting lead in water. Result indicated that a highly efficient lead removal performance with 95% of lead eliminated within 5 min. The adsorption capacity of lead was 8.2 times than that of AC support only. Pb(II) was converted to insoluble forms. These reactions were accompanied by the surface oxides aging of NZVI/AC. There was no iron release during the processes of lead removal. This study strengthens the understanding of the characteristics of supported nanoscale zero-valent iron and mechanisms for lead conversion, and it also broadens its application field. From practical application point of view, lead ions are not only adsorbed, but also further converted into low-toxicity or nontoxic substances.
The research was published in the Journal of Hazardous Materials 2019, 361, 37-48 (https://doi.org/10.1016/j.jhazmat.2018.08.082), and titled as “Performance of Pb(Ⅱ) removal by an activated carbon supported nanoscale zero-valent iron composite at ultralow iron content”. The first author is Liu Xuejiao and the corresponding author is Pro.Wang Yin. This research was supported by the China-Japan Research Cooperative Program and the National Natural Science Foundation of China, etc.
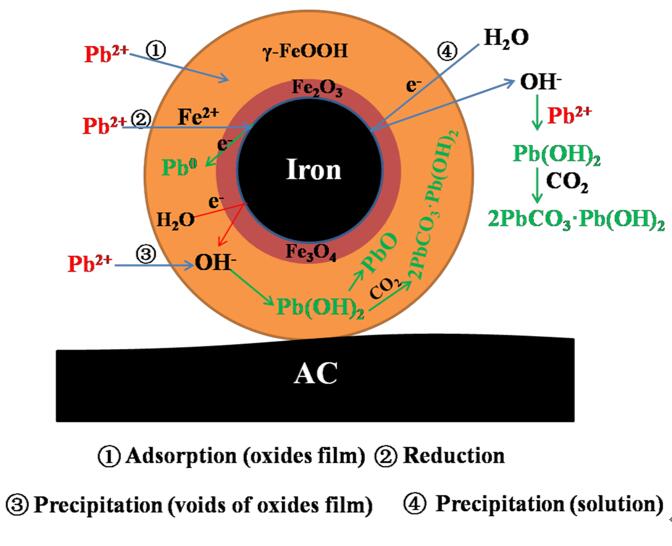
Figure 1. Mechanism for the Pb(Ⅱ) removal by NZVI/AC composite