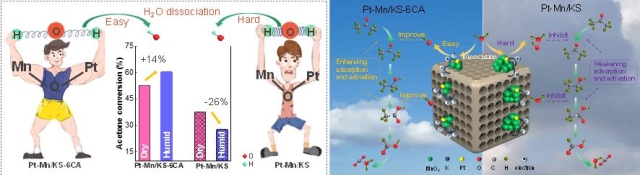
As a generally existing component in industrial streams, H2O usually inhibits the catalytic degradation efficiency of volatile organic compounds (VOCs) greatly. Here, we propose a novel strategy that accelerates the H2O dissociation and facilitates positive feedbacks during VOC oxidation by fabricating citric acid (CA)-assisted Pt(K)-Mn2O3/SiO2 (Pt-Mn/KS-xCA). Results reveal that the complexation of carboxyl groups of citric acid with Mn cations leads to the formation of small Mn2O3 (4.1 ± 0.2 nm) and further enhances the Mn-O-Pt interaction (strengthened by the Si-O-Mn interaction), which can transfer more electrons from Pt–Mn/KS-6CA to H2O, thus facilitating its breaking of covalent bonds. It subsequently produces abundant surface hydroxyl groups, improving the adsorption and activation abilities of acetone reactant and ethanol intermediate. Attributing to these, the acetone turnover frequency value of Pt–Mn/KS-6CA is 1.8 times higher than that of Pt-Mn/KS at 160 °C, and this multiple changes to 6.3 times in the presence of H2O. Remarkably, acetone conversion over Pt–Mn/KS-6CA increases by up to 14% in the presence of H2O; but it decreases by up to 26% for Pt–Mn/KS due to its weak dissociation ability and high adsorption capacity toward H2O. This work sheds new insights into the design of highly efficient catalytic materials for VOC degradation under humid conditions.